
KineMatch® Patient-Matched PFR
Technology your patients can appreciate
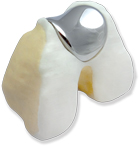 Designed specifically for the small but challenging group of patients with isolated, end-stage patello-femoral disease, the KineMatch PFR (Patello-Femoral Replacement) offers a uniquely effective and conservative resurfacing solution. Because the device is precisely pre-fitted to the patient’s anatomy using CT (Computed Tomography) data, no resection of femoral bone is required. This also ensures proper implant alignment and preserves bone stock for the future. The KineMatch® patella, a standard all-polyethylene button, is used in conjunction with the femoral component.
Designed specifically for the small but challenging group of patients with isolated, end-stage patello-femoral disease, the KineMatch PFR (Patello-Femoral Replacement) offers a uniquely effective and conservative resurfacing solution. Because the device is precisely pre-fitted to the patient’s anatomy using CT (Computed Tomography) data, no resection of femoral bone is required. This also ensures proper implant alignment and preserves bone stock for the future. The KineMatch® patella, a standard all-polyethylene button, is used in conjunction with the femoral component.
And because the goal is not only to reduce pain but also to improve function, the KineMatch PFR is designed to restore knee kinematics by rebuilding the patellar groove for improved patellar tracking and by restoring the A-P position of the patella for proper quadriceps function.
The device is implanted in a simple surgical procedure with minimal joint disruption. A drill template matching the patient-specific femoral implant in both femoral fit and perimeter shape is provided for each case. The drill template margin is marked and the cartilage is removed from the bone in the area under the femoral component. Three peg holes are drilled in the anterior distal femur using the template. The CoCr femoral and UHMWPe patella implants are fixed using bone cement.
Femoral components thickness is kept to a minimum in order to replicate normal cartilage thickness and provide for normal joint kinematics.
Benefits of KineMatch® Patello-Femoral Replacement
• Created to address intractable patello-femoral disease when other treatment options have failed.
• Designed to approximate normal kinematics by re-establishing trochlear groove alignment and depth with the aim of simulating the function of a normal anatomic sulcus to facilitate proper patella tracking.
• Avoids femoral bone resection by utilizing CT modeling technology to achieve an individual custom fit to the patient’s distal anterior femoral anatomy.
• Custom fit minimizes problems of soft tissue impingement often seen with “off-the-shelf” patello-femoral implant designs.
• Post-operative knee function is potentially better compared to total knee arthroplasty.
• Generally, operative time is significantly less than total knee surgery.
• Simple and Fast Surgical Technique
• May reduce post-operative pain, morbidity, and physical therapy requirements as compared to alternate treatment options.
Surgeon Testimonials
“We formally reviewed our experience with custom patellofemoral arthroplasty and found good short-term functional outcomes and low revision rates with very few complications. My patients with isolated patellofemoral arthritis who have failed nonoperative management benefit from KineMatch custom PFA.”
Tarun Bhargava, MD, Board Certified, Fellowship Trained Orthopaedic Surgeon, Wichita, KS, USA
“KineMatch is the perfect solution for my patients with end-stage patellofemoral arthritis. These patients tend to be younger than typical total knee replacement candidates, and are looking to return to an active lifestyle. With KineMatch, there is no bone resection, which makes it a conservative, simple and fast surgery. Every KineMatch implant is custom made so it is always a perfect fit!”
Peter W. Callander, MD, Board Certified, Fellowship Trained Orthopaedic Surgeon, San Francisco, CA, USA
“I have tried several of the off-the-shelf patello-femoral arthroplasty implants and have concluded that the KineMatch Patient-Matched PFR is consistently the best option for my patients requiring patellofemoral arthroplasty. The concept of using CT data to customize the backside of the implant to intimately fit the trochlea without need for bone resection is conservative, effective, and simplifies and speeds the surgical procedure.”
Russell Cecil, MD, Mohawk Valley Orthopedics, Amsterdam, NY, USA
“I have been performing KineMatch custom-fit patello-femoral arthroplasty for years. I am very pleased with the rapid return of function and range of motion.”
Gregg R. Foos, MD, Double Board Certified, Fellowship Trained Orthopaedic Surgeon, Tinton Falls, NJ, USA
“I began using the KineMatch PFR in 2011 and have performed over 90 cases to date. I have found this device to be an excellent solution for patients with isolated end-stage patello-femoral disease.”
Robert J. Greenhow, MD, FRCSC, Diploma Sports Medicine, Peak Orthopedics and Spine (a division of Orthopedic Centers of Colorado) Englewood, CO, USA
“I started performing KineMatch custom-fit patello-femoral arthroplasty in 2007. I am very pleased with the rapid pain relief, quick return of range of motion and function, as well as the short operative time and learning curve.”
Ronald P. Grelsamer, MD, Chief of Patello-Femoral Reconstruction Mount Sinai Medical Center, New York, NY, USA
“In my experience the KineMatch PFR has been an excellent option for younger (<55 year old) patients with end-stage OA isolated to the PF joint. We have used the KineMatch system with success on more than 60 patients in our practice since 2007. With proper patient selection this has been a reliable and powerful tool to restore quality of life, even in individuals with very active lifestyles.”
Jon Henry, MD, Aurora BayCare Orthopedic & Sports Medicine Center, Green Bay, WI, USA
“This patient specific design and manufacturing technique ensures accurate and precise anatomic fit while simultaneously providing proper patellofemoral alignment and medial lateral constraint.”
Adolph V. Lombardi, Jr., MD, FACS, Joint Implant Surgeons, Inc. New Albany, OH, USA
“The KineMatch device has offered a remarkable benefit and return to function for a number of my patients with intractable patello-femoral disease who were otherwise facing the prospect of TKR.”
Domenick J. Sisto, MD, Sherman Oaks, CA, USA
“I have found the KineMatch patella femoral replacement system offers excellent results in patients with arthritis isolated to the patello-femoral (kneecap) portion of the knee. It works especially well for younger active patients who don’t warrant a total knee replacement.”
Brian M. Torpey, MD, Double Board Certified, Fellowship Trained Orthopaedic Surgeon, Tinton Falls, NJ, USA
Videos
Lombardi KineMatch Patient-Matched PFR Surgical Video
KineMatch Patellofemoral Replacement System
A KineMatch Patient’s Personal Story
KineMatch PFR 3D Animation
Patents
US Patent Nos. 6,712,856, 6,905,514, 7,517,365, 7,935,150, 8,419,741, 8,771,281, 8,936,601, 8,936,602, 8,961,529, 9,393,032. Europe Patent Nos. 1,265,559 and 3,181,068.
